Gosselies, Belgium – April 9th, 2026
EPICS Therapeutics announces that new preclinical data on EP102, an emerging highly promising METTL3¹ inhibitor, will be presented at the American Association for Cancer Research Annual Meeting 2026, in San Diego, California on April 22, 2026.
The abstract, titled “EP102: pharmacological inhibition of METTL3 is effective as a monotherapy and potentiates radiation therapy in pancreatic adenocarcinoma via DNA damage and stemness modulation,” highlights the therapeutic potential of EP102 in pancreatic ductal adenocarcinoma (PDAC) one of the most aggressive cancers, where five‑year survival remains close to 10%.
Highlights from the preclinical findings include:
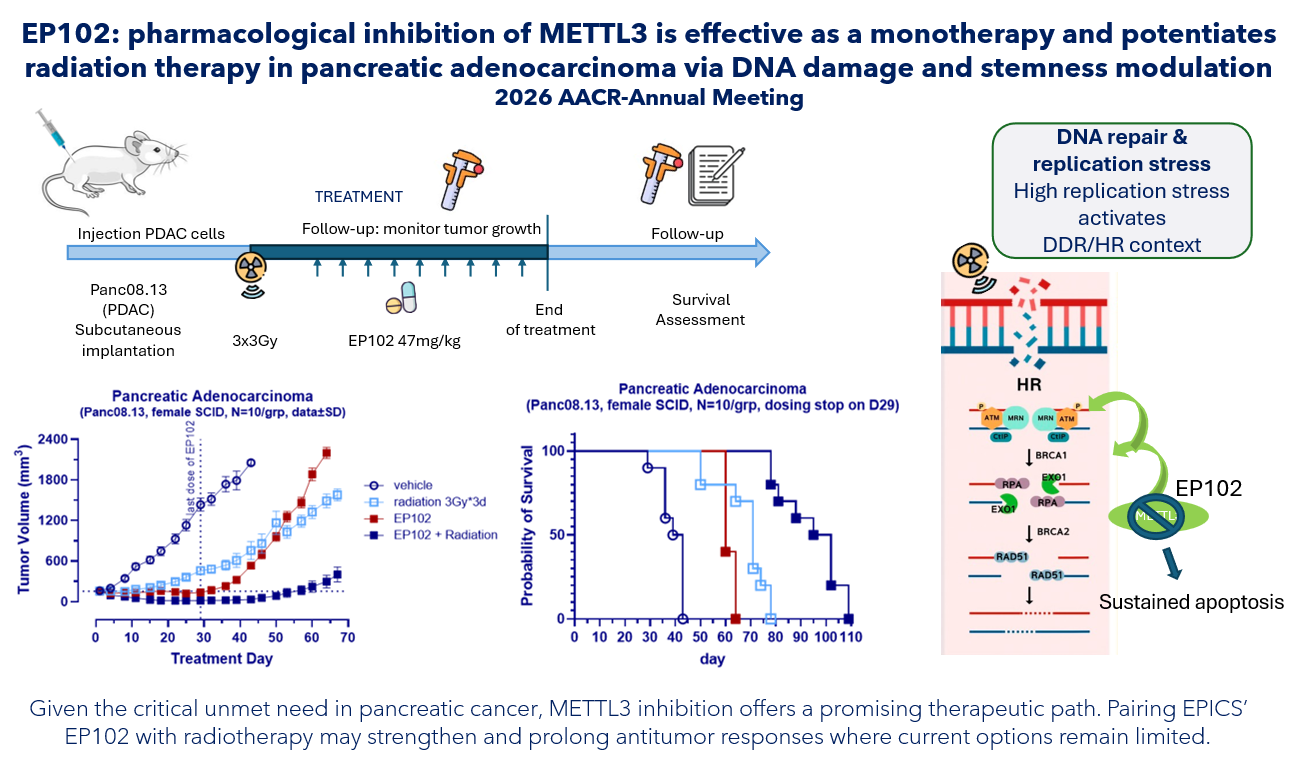
- Strong monotherapy activity: EP102 demonstrated significant, dose-dependent tumor growth inhibition in a PDAC xenograft model².
- Enhanced effect with radiotherapy: When combined with radiation therapy, EP102 induced marked tumor shrinkage and extended treatment durability, supporting its robust synergistic potential.
- Mechanism of action – increased DNA damage: EP102 reduced the expression of key DNA repair and survival genes, inducing accumulation of DNA damage response. These effects were further amplified in combination with radiotherapy.
- Marked induction of tumor cell death: EP102, particularly in combination with radiotherapy, triggered pronounced apoptosis (programmed cell death) and impaired homologous recombination repair (a key pathway cells use to accurately fix DNA damage), resulting in significant tumor regression.
- Favorable tolerability profile: EP102 was well tolerated as a monotherapy in preclinical studies, confirming its potential safety in further development.
These findings suggest that METTL3 inhibition disrupts m⁶A-dependent DNA repair and survival pathways, rendering tumor cells more vulnerable to radiation-induced damage and enhancing the antitumor response.
Commenting on the findings, Graeme Fraser, PhD, Chief Executive Officer and Chief Scientific Officer, said: “Pancreatic cancer is a devastating disease with limited treatment options, but where radiation therapy does offer some benefit. Our preclinical data demonstrates that EP102 greatly improves the durable response to radiation through synergistic effects on tumoricidal DNA damage response pathways, complemented by an additional effect of EP102 to reduce cancer stem cells and thereby prevent relapse. EP102 is currently being evaluated in a clinical trial where we would expect this program to deliver meaningful benefit to patients.”
Providing further scientific context, Andrea Casazza, PhD, Head of Cancer Biology, added: “Pancreatic cancer is a silent adversary: emerging late, agile in its ability to evade the immune system, shielded by dense fibrotic tissue, and adapting faster than most treatments can respond. By targeting METTL3, EP102 aims to disrupt the cancer’s core survival programs while enhancing the effects of radiation therapy, amplifying DNA damage and weakening tumor adaptability. Together, these effects may restore treatment sensitivity, limit tumor regrowth, and potentially deliver more durable outcomes for patients.”